 Dalton's law – Gas law describing pressure contributions of component gases in a mixture.This forms a pressure difference between the air inside the lungs and the environmental air pressure, which in turn precipitates either inhalation or exhalation as air moves from high to low pressure. This commonly involves explaining how the lung volume may be increased or decreased and thereby cause a relatively lower or higher air pressure within them (in keeping with Boyle's law). The three gas laws in combination with Avogadro's law can be generalized by the ideal gas law.īoyle's law is often used as part of an explanation on how the breathing system works in the human body. Here P 1 and V 1 represent the original pressure and volume, respectively, and P 2 and V 2 represent the second pressure and volume.īoyle's law, Charles's law, and Gay-Lussac's law form the combined gas law. 
Mathematically, Boyle's law can be stated as: 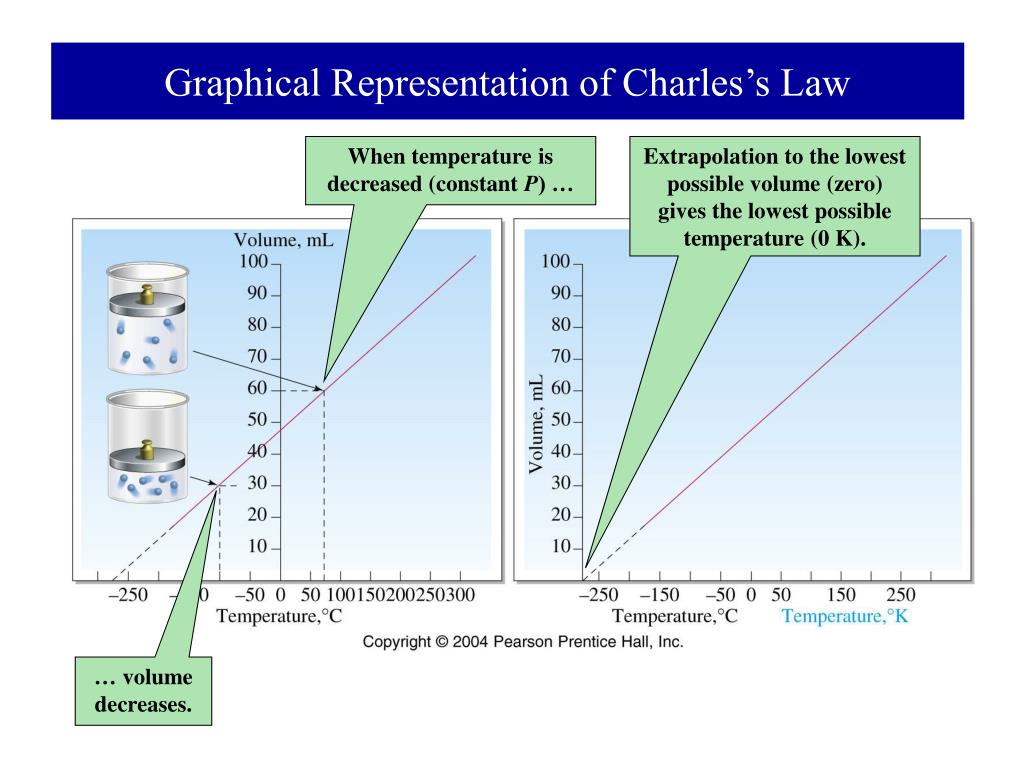
The absolute pressure exerted by a given mass of an ideal gas is inversely proportional to the volume it occupies if the temperature and amount of gas remain unchanged within a closed system. 
We know the value of your time and strive hard to deliver the best and invest in it with precision.Boyle's law, also referred to as the Boyle–Mariotte law, or Mariotte's law (especially in France), is an experimental gas law that describes the relationship between pressure and volume of a confined gas. In Middle-East and Singapore, we are the among the most recommended educational sites for an online home tutor at very lowest tuition fees. Vedantu Classes rank as the best LIVE and Online Tutoring Website in top ten cities of India- Bangalore, Delhi, Hyderabad, Chennai, Mumbai, Patna, Kolkata, Pune, Chandigarh and Jaipur. FREE Sample Papers and Important questions are extracted, solved and discussed, ensuring that you are 100% prepared before any exam. To promote talent and potential, the Prices for Master Classes are very affordable. Master Teachers cater to teaching Maths, Physics, Chemistry and Biology (Science) for 6th to 12th grades across CBSE and ICSE Boards. To ensure that motivation is stirred in the best proportion for your clear understanding, a good number of quizzes and Objective tests like V-Brainer, V-Maths, Turbo Maths are organized to impart knowledge and reward the best performers with surprise gifts. Hours and Hours of Study with no fun, is a bad idea for you, foreseeing the long run. Vedantu is the first choice of students aspiring to score full marks in their ICSE and CBSE Board exams or to crack any competitive exam like IIT JEE (Mains & Advanced), Kishore Vaigyanik Protsahan Yojana (KVPY), National Talent Search Exam (NTSE), International Math Olympiad (IMO), International English Olympiad (IEO). Interactive approach establishes a well-deserved academic connect between you and Master Teachers. Sessions get recorded for you to access for quick revision later, just by a quick login to your account. Your academic progress report is shared during the Parents Teachers Meeting. Assignments, Regular Homeworks, Subjective & Objective Tests promote your regular practice of the topics. Revision notes and formula sheets are shared with you, for grasping the toughest concepts. WAVE platform encourages your Online engagement with the Master Teachers. We provide you year-long structured coaching classes for CBSE and ICSE Board & JEE and NEET entrance exam preparation at affordable tuition fees, with an exclusive session for clearing doubts, ensuring that neither you nor the topics remain unattended. 
We have grown leaps and bounds to be the best Online Tuition Website in India with immensely talented Vedantu Master Teachers, from the most reputed institutions. Vedantu LIVE Online Master Classes is an incredibly personalized tutoring platform for you, while you are staying at your home. To answer questions related to Charles law, you need to focus on clarifying your doubts. Gas laws are defined at idea conditions and are then utilized in real-time situations for making proper calculations. When the temperature decreases, the volume will decrease, and vice versa.Īccording to the above statement, we can write the following when comparing a substance in two different circumstances: His unpublished work provides extensive details about this law.Ī detailed explanation of how gas expands when the temperature rises is provided by Charles' Law, also known as the law of volumes. According to the law, when constant pressure is applied to a sample of dry gas, the Kelvin temperature, and the volume will be directly proportional.Ī French physicist named Jacques Charles first proposed this law in the year 1780. As the temperature and pressure of an ideal gas remain constant, Charles' law states that the volume is directly proportional to its absolute temperature at constant pressure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
